Research
Identifying the Biosynthetic Pathways for Pharmaceutical alkaloids in Mitragyna species
Swathi Nadakuduti, Principal Investigator on the USDA-NIFA grant, in her greenhouse facility along with her PhD candidate, Larissa Laforest (in orange) from Plant Molecular and Cell Biology Program.
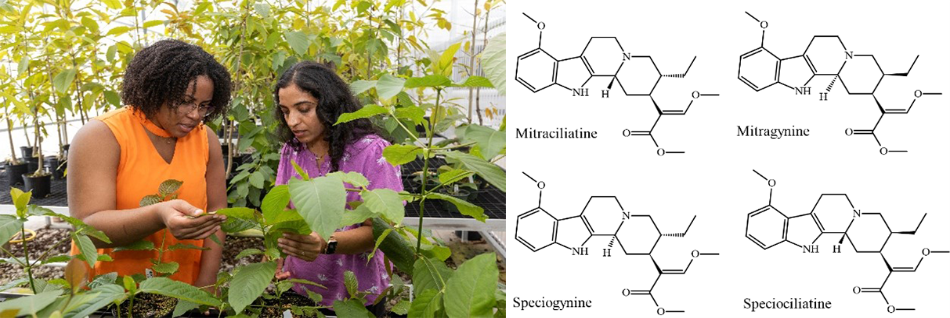
A multidisciplinary team of researchers from UF/IFAS, College of Pharmacy and UBC, Canada will work on pathway discovery of pharmaceutical alkaloids in kratom. The leaves of Mitragyna speciosa (kratom), produce more than 50 monoterpene indole alkaloids (MIAs) and spirooxindole alkaloids associated with varied pharmaceutical uses. The MIA mitragynine has shown promise as a potential treatment for pain, opioid use disorder, and opioid withdrawal without any demonstrated addiction potential; and the spirooxindole mitraphylline shows anti-tumor activity. The existing knowledge gap in the biosynthesis of bioactive M. speciosa alkaloids prevents systematic advances in engineered production of these high value compounds, creating a bottleneck for pharmaceutical research and production. A multi-faceted approach incorporating genomics, transcriptomics, and metabolomics will be jointly applied to enable pathway discovery of diverse bioactive alkaloids with the goal of supporting synthetic biology efforts to produce these alkaloids in appreciable quantities for clinical study.
Exploring Florida Native plants for metabolites of pharmaceutical value
We are interested in exploring medicinally relevant species of Florida natives for plant-specialized metabolites to identify bioactive compounds, their spatio-temporal patterns of accumulation and decipher metabolic pathways.
Scutellaria, a Florida native ornamental with potential anti-cancer properties
Scutellaria spp has tremendous potential for the ornamental and medicinal plant industry in Florida. The root extract of S. baicalensis has been widely used for medicine in Asia for over 2,000 years, and it has a variety of bioactivities, e.g., anti-inflammatory, anti-cancer and neuro-psychologic properties due to synthesis of unique flavonoids and indolic compounds. Commercially, it has a 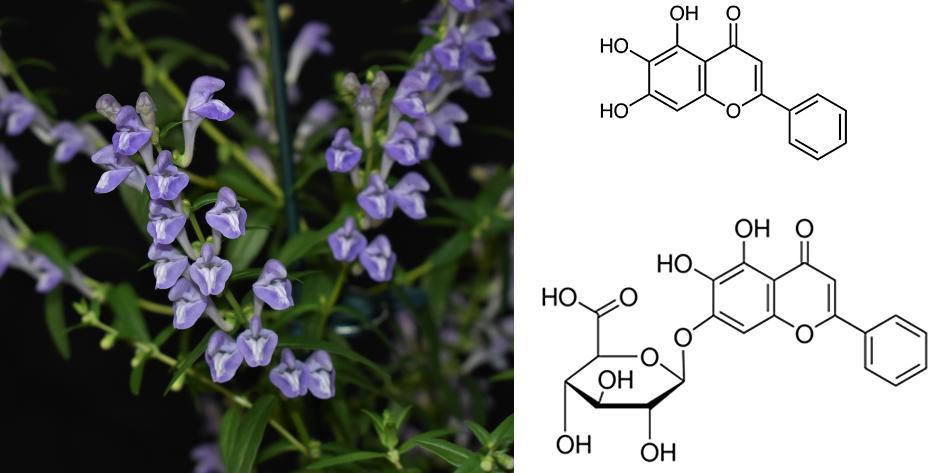 market for ornamental and medicinal applications including supplement capsules, dried plants for tea, powdered or liquid extracts. Florida native species, S. integrifolia and S. arenicola also synthesize these active medicinal compounds and in order to develop Scutellaria crop as a new enterprise crop, we need to identify germplasm and best production practices, while evaluating the metabolite profiles under controlled environments to maximize the synthesis of active pharmaceuticals. This project is a part of a new UF team “MICE (Medicinals In Controlled Environments)” across UF IFAS and College of Pharmacy
market for ornamental and medicinal applications including supplement capsules, dried plants for tea, powdered or liquid extracts. Florida native species, S. integrifolia and S. arenicola also synthesize these active medicinal compounds and in order to develop Scutellaria crop as a new enterprise crop, we need to identify germplasm and best production practices, while evaluating the metabolite profiles under controlled environments to maximize the synthesis of active pharmaceuticals. This project is a part of a new UF team “MICE (Medicinals In Controlled Environments)” across UF IFAS and College of Pharmacy
As a part of “Support for Emerging Enterprise Development Integration Teams” (SEEDIT), we will evaluate Florida native Scutellaria species in FL landscape locations for ornamental & medicinal purposes, manipulate controlled environment in order to maximize pharmaceutically active metabolites, quantify price and quality of existing Scutellaria products and develop enterprise budget and communicate project outcomes with producers, processors and entrepreneurs.
Saw palmetto, a Florida native palm used as a largest herbal supplement globally

Saw palmetto, Serenoa repens is a Florida native palm and the berries are wild harvested throughout the state of FL. Saw palmetto is the third most utilized dietary supplement in the US for benign prostatic hyperplasia and lower urinary tract symptoms. S. repens grows in a variety of habitats, including sandy coastal lands, pine flatwoods, and under story areas in hardwood hammocks in FL. We are interested in exploring the genetic diversity and correlate with chemical diversity (lipido-sterolic composition) to understand the pharmaceutical potential of berries across the FL geography.
Dissecting molecular mechanisms to enhance fruit photosynthesis and nutritional quality of tomato
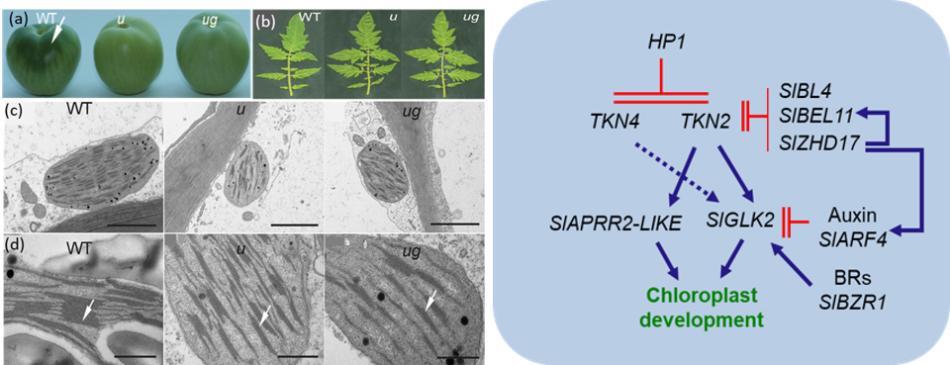
Swathi Nadakuduti, Principal Investigator on the USDA-NIFA grant is working on improving the fruit nutritional quality using tomato as a model system. Although leaves are the primary photosynthetic organs, up to 20% of the carbon in tomato fruit is derived from fruit photosynthesis itself, impacting yield, nutrient content, flavor, and overall fruit quality. PI's research identified TKN4 in tomato, a Class I KNOTTED-like homeobox (KNOX) gene that also influences chloroplast development and chlorophyll accumulation in fruit. Tomato uniform ripening (u) and uniform grey green (ug) mutants showing differential chlorophyll accumulation will be used in identifying the KNOX regulatory network that has the long-run potential to favorably engineer photosynthesis contributing to yield and quality of food crops.
Genome-wide evaluation of off-targets and phenotypic effects in genome-edited citrus for resistance to Huanglongbing and citrus canker
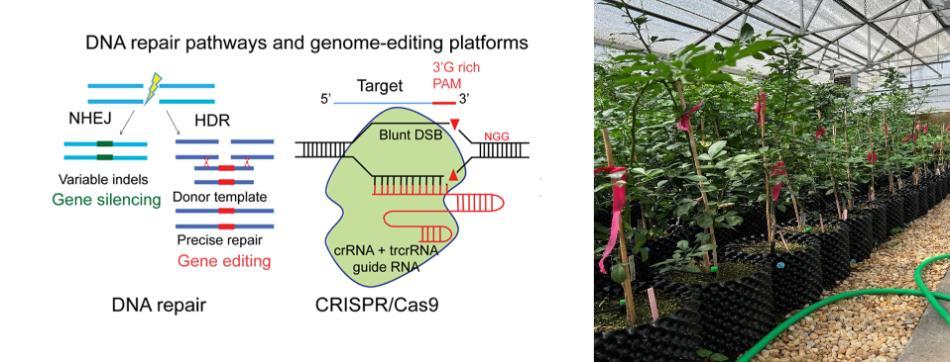
Citrus is one of the most important fruit crops in the U.S. and in the world with many cultivated varieties. The production of citrus in the U.S. has been devasted by the bacterial diseases Huanglongbing (HLB), and to some extent by citrus canker. We have generated multiple citrus genome-edited lines targeting six selected host plant susceptibility (S) genes to achieve resistance against these devastating bacterial diseases. Because commercial citrus has to be clonally propagated, gene-edited citrus plants will have stably incorporated CRISPR reagents in their genomes through generations. This indicates that there is a possibility of continued CRISPR activity affecting both genotype and phenotype of gene-edited citrus lines. Therefore, risk-assessment of gene-editing technology for engineering citrus species is imperative to meet the urgent need from the U.S. citrus industry. We propose to perform comprehensive evaluation of genome-wide off target effects and associated phenotypes in these citrus gene-edited lines compared to non-modified controls in field studies. Further, we will also evaluate the stability of edits and phenotypes over multiple years under field conditions. The results from this project will assist regulatory agencies to effectively and comprehensively assess the risks of gene-editing in citrus at both genotypic and phenotypic levels.
